 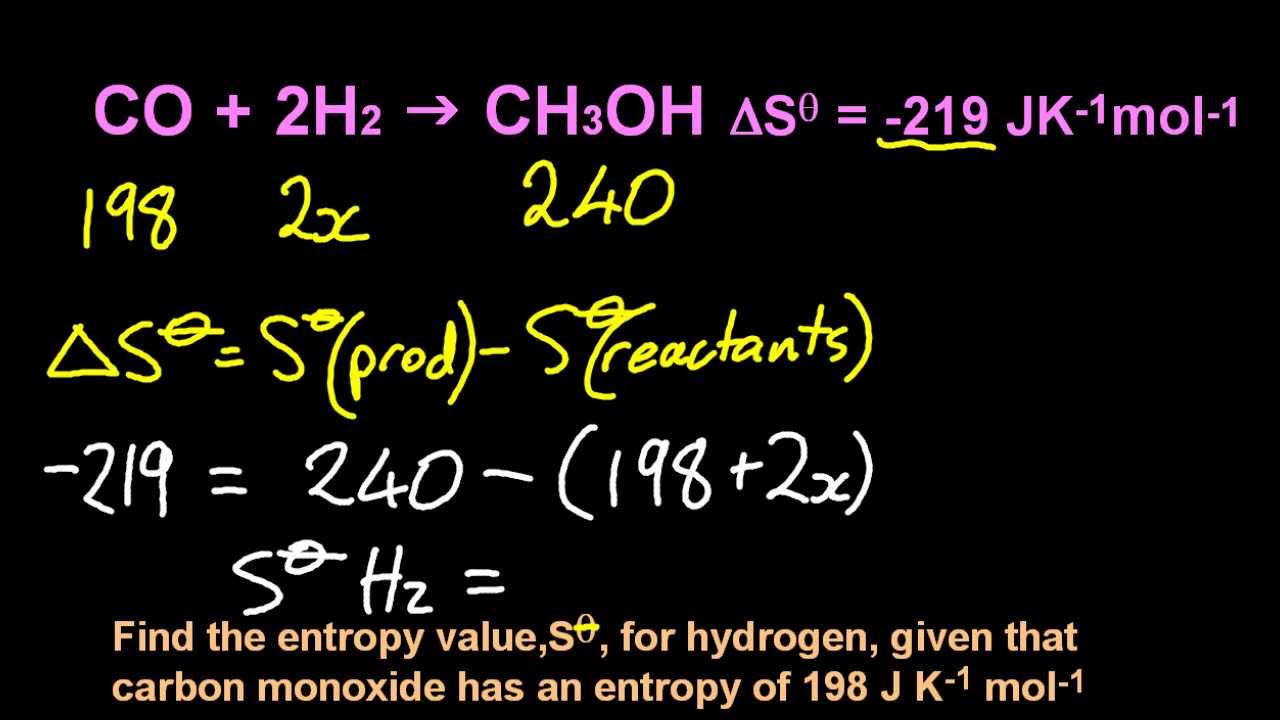
If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive. 
T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). As T increases, the T∆S component gets bigger. ∆H is still positive and ∆S is still whatever sign you figured out above. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again. From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. Temperature is always positive (in Kelvin). We know (from the question) that ∆G is negative and that ∆H is positive. The only is important it means without any other changes occurring. The second law thus tells us that we cannot get work from a single reservoir only. Because entropy is a state function, however. The change in entropy that accompanies the conversion of liquid sulfur to S (S fus() S 3 in the cycle) cannot be measured directly. The total entropy change in the proposed process is thus less than zero, which is not possible. The transition from S to S can be described by the thermodynamic cycle shown in part (b) in Figure 19.3.3, in which liquid sulfur is an intermediate. Where q is the heat transferred in joules and ΔT is the change in temperature in Kelvin.This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S. Figure 5.5: Work from a single heat reservoir. If there are 2 moles of reactant forming 1 mole of product, the number of states possible decreases and entropy decreases, likewise, if 1 mole of reactant forms 2 moles of product, the entropy increases.Įntropy can be calculated using the equation Example of increasing entropy: Ice melting in a warm room is a common example of increasing entropy. 
If a substance goes from a liquid to a solid, the molecules in the system occupy a lower number of states (think about the decrease of molecule activity in water when it freezes to ice) and therefore the entropy decreases if a substance goes from a solid to a liquid, the entropy increases. When considering whether reactions have an increase or decrease in entropy, two factors are important: The number of moles created and states of the reactants versus the products. Liquids can be considered the second most disordered, and solids can be considered the least disordered. Because molecules in gases are able to change both volume and shape, they can be considered the most disordered. We can think of the degrees of the number of different states the molecules in each can occupy. Solids, liquids, and gasses all have different degrees of disorder. The concept of disorder can best be described in terms of the states of matter. 
The unit of entropy is Joules per Kelvin or J/K.Įntropy is the measure of the disorder of a system and can is the energy of a system over its temperature, represented as J/K. The Second Law of Thermodynamics states that the entropy (or disorder) of a system is constantly increasing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
